Company millestone: Milla Pharmaceuticals Receives FDA Approval for Dexmedetomidine Hydrochloride Injection – Alter Pharma
Slayback Pharma announces approval and immediate launch of Dexmedetomidine Hydrochloride in 0.9% Sodium Chloride Injection in 200 mcg/ 50 mL and 400 mcg/ 100 mL (4 mcg/mL) vials, generic equivalent of Precedex™ 4 mcg/ mL | BioSpace

Dexmedetomidine HCl, Preservative Free / 0.9% Sodium Chloride 100 mcg / mL Injection Single-Use Vial 25 x 2 mL - Merit Pharmaceutical
Dexmedetomidine Hydrochloride Injection Manufacturer,Dexmedetomidine Hydrochloride Injection Exporter,Supplier

Milla Pharmaceuticals Inc. and the Alter Pharma Group Announce Yet Another "First Cycle" FDA Approval for an Abbreviated New Drug Application (ANDA), Dexmedetomidine Hydrochloride Injection

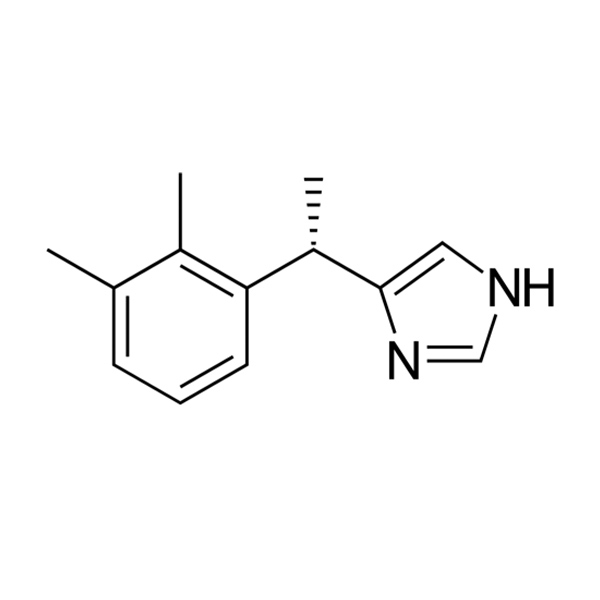
emDOCs.net – Emergency Medicine EducationDexmedetomidine, almost as many uses as there are letters - emDOCs.net - Emergency Medicine Education

Revertidine (Atipamezole Hydrochloride) Sterile Injectable Solution 5mg/mL (10 mL) | On Sale | EntirelyPets Rx
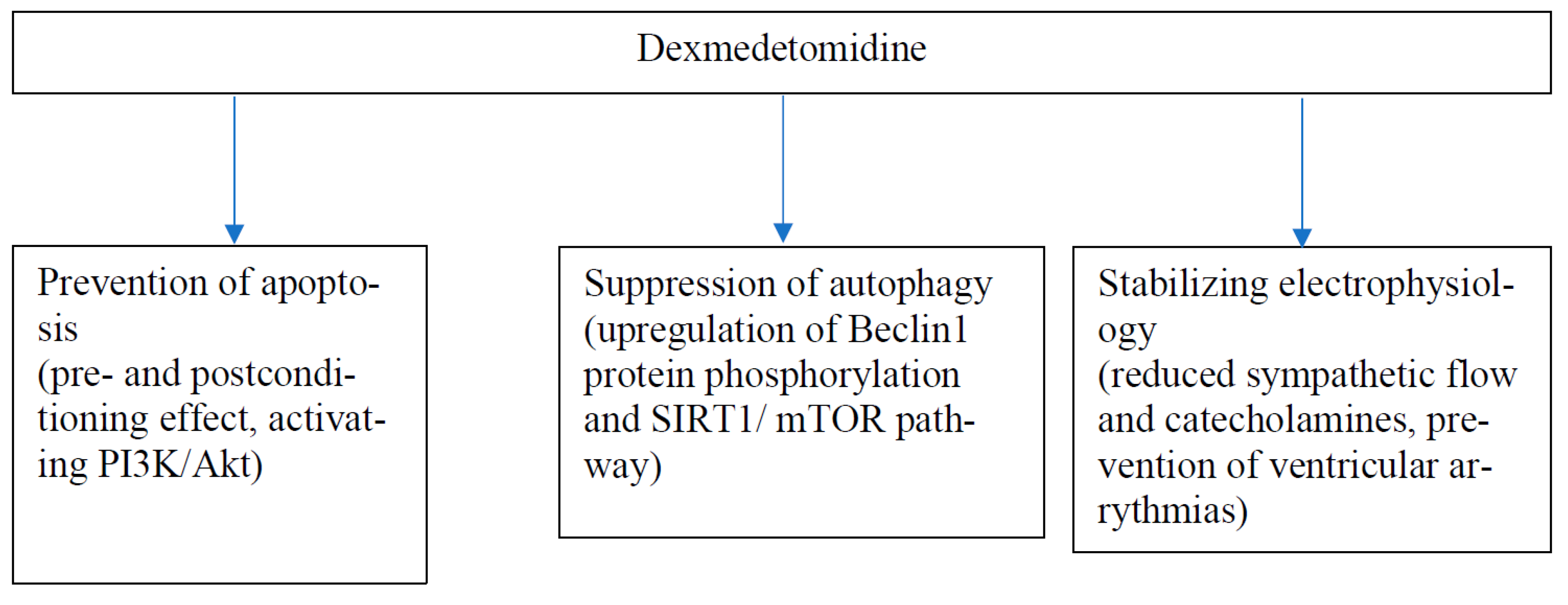
Pharmaceuticals | Free Full-Text | Effects of Dexmedetomidine on Basic Cardiac Electrophysiology in Adults; a Descriptive Review and a Prospective Case Study
Fresenius Kabi Introduces Dexmedetomidine Hydrochloride in 0.9% Sodium Chloride Injection - Fresenius Kabi USA